
AI in Drug Development Enabling Diseases Prevention
Genix.ai supports pharma and biotech teams with AI-assisted genomic intelligence to accelerate biomarker discovery, improve cohort stratification, and enable data-driven decisions across the drug development lifecycle.

The Challenge in Pharma & Biotech Genomics
Pharma and biotech organizations generate vast amounts of genomic
and clinical data—but extracting value remains difficult

How Genix.ai Supports Pharma & Biotech Teams
Genix.ai provides a research- and development-ready genomics intelligence platform that bridges discovery, translation, and clinical application.

Analyze genomic data with clinical

population context

Identify and validate biomarkers

Build reproducible, explainable

Support decision-making across R&D Clinic

AI-powered faster discovery

Your genes have answers. Are you ready?
key hospital use cases
key hospital use cases
Biomarker Discovery & Validation
Patient & Cohort Stratification
Translational Research
Clinical Development Support
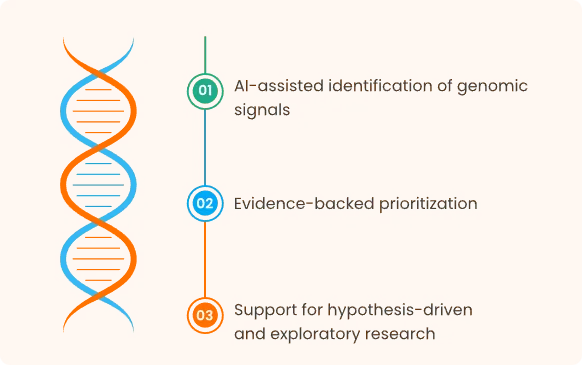
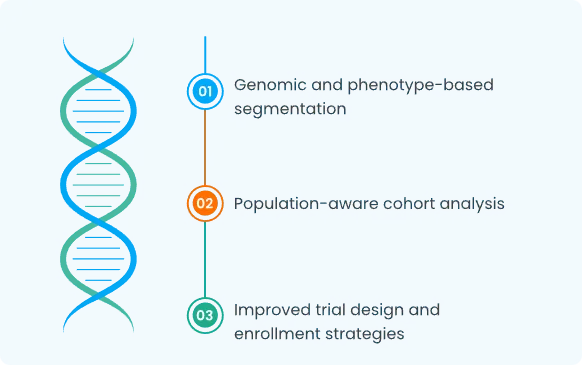
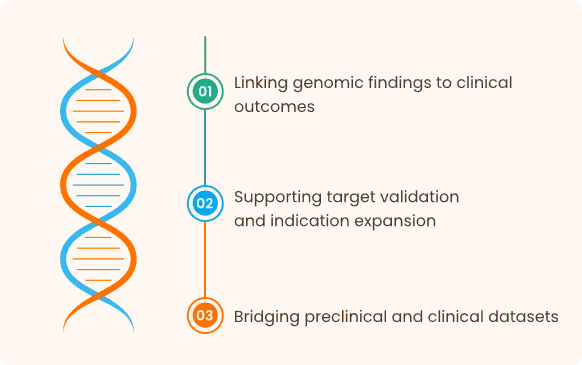
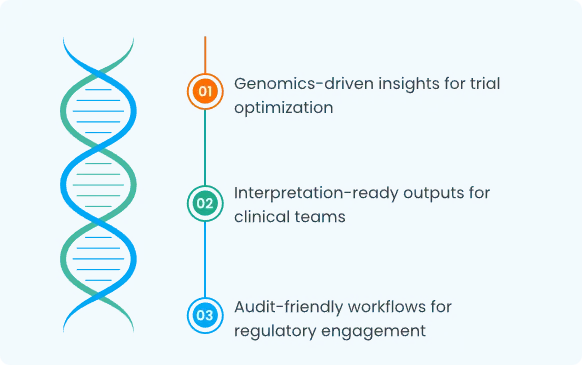
How the Platform Fits into R&D Workflows
Genix.ai provides a research- and development-ready genomics intelligence platform that bridges discovery, translation, and clinical application.
Data Integration
Step 1
- Secure ingestion of genomic and phenotypic data
- Support for structured and semi-structured datasets
Analytical Processing
Step 2
- Bioinformatics and feature extraction workflows
- AI-assisted prioritization layers
Interpretation & Review
Step 3
- Explainable evidence aggregation
- Expert review and refinement
Insight Delivery
Step 4
- Research-ready datasets
- Decision-support summaries
- Exportable outputs and APIs

BUILT FOR SCIENTIFIC RIGOR AND REPRODUCIBILITY
Genix.ai is built with hospital realities in mind
Transparent AI-assisted suggestions
Versioned workflows and annotations
Clear separation of AI suggestions and expert conclusions
Reproducible pipelines
Security, Privacy & Collaboration Readiness
Pharma and biotech organizations trust Genix.ai because the platform supports

Secure collaboration across internal and external teams
-2.svg)
Controlled access to sensitive datasets
-3.svg)
Deployment flexibility (Private VPC and On-Premise preferred)
-4.svg)
Alignment with data governance

-5.svg)
compliance requirements
Deployment options for hospitals
Hospitals can choose the model that best fits their policy and risk posture
SaaS
For early-stage exploration and pilots
On-Premise
For sensitive or regulated datasets
Private VPC
Preferred for R&D and clinical programs
Why Pharma & Biotech Teams Choose Genix.ai
Accelerated biomarker discovery
Improved patient stratification
Explainable, audit-ready AI
Scalable from research to clinical programs
Designed for long-term R&D partnerships

Who Uses Genix.ai in Pharma & Biotech
Translational medicine teams
Biomarker and genomics scientists
Clinical development leaders
Data science and informatics teams
External research collaborators

Start a Conversation
Share a few details and our team will connect with you
Frequently Ask Questions
1. How does Genix.ai accelerate the biomarker discovery and validation process?
2. Can the platform support patient stratification for clinical trials?
3. How do you ensure the reproducibility of R&D pipelines?
4.Does the platform integrate with existing bioinformatics tools and pipelines?
5. What deployment models are available for sensitive IP and clinical data?
- Private VPC: A dedicated, isolated cloud environment.
- On-Premise: For highly regulated data where sovereignty is mandatory.
- SaaS: For early-stage exploration and pilot programs.
6. How do you handle "Black Box" concerns in AI modeling?
7. Can Genix.ai handle multi-omic datasets beyond genomics?
8. Is the platform ready for regulatory audits and GxP compliance?

Bring Clinical Genomics into Routine Hospital Care
If your hospital or health system is evaluating how to operationalize genomics responsibly and at scale, Genix.ai can support your journey.








